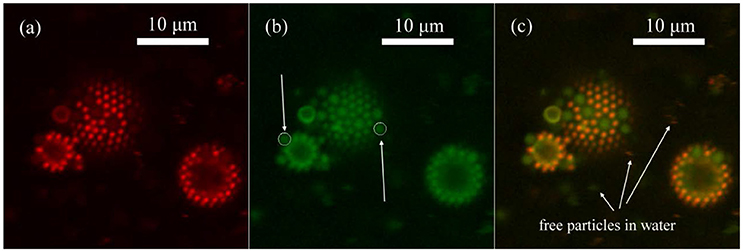
However, to enable the rational design of responsive emulsions, the origin of the destabilisation cannot remain a mystery and must be understood. With these insights, the previously established direct destabilisation mechanism is brought into question 28, 29. Ellipsometry 44, neutron reflectometry 45 and molecular dynamic simulations 43, 45, 46 suggest that only the part of the microgel exposed to the water phase changes with temperature and collapses. Additionally, the lateral dimensions of the microgels did not change upon heating and, therefore, there were no changes in the interfacial assembly 43, 44. Two recent studies of microgel monolayers showed that they persisted throughout temperature cycling and that no desorption occurred 43, 44. It was also proposed that microgels desorbed from the oil/water interface, again lowering coverage 30, 36, although this did not appear in all reported scenarios 38, 39, 42. It was speculated that when heated above T VPT, the stabilizing microgels shrink laterally and the reduced interfacial coverage destabilizes the emulsions 29, 30, 31, 33, 34, 35, 36, 37 with a potential change in the mechanical properties of the interfacial microgel monolayer 38, 39, 40, 41. Earlier reports attributed the breaking directly to the volume phase transition of the stabilizing microgels. The fundamental mechanism behind the rupture of such oil in water emulsions is, 15 years after its discovery 30, 31, still under debate 28, 29. As a result, emulsions stabilized by PNIPAM microgels are stable at room temperature, but can destabilize above T VPT 24, 25, 26, 27, 28, 29, 30, 31, 32. Frequently used stabilizers in thermo-responsive emulsions are poly(N-isopropylacrylamide) (PNIPAM) microgels particles, which transition from a swollen to a collapsed state above their volume phase transition temperature ( T VPT) of 32 ☌. E.g., this can occur through modifying the continuous phase via a change of pH 13, 14, 15, 16, 17, the addition of sugar 18 or oxidizing agents 19, the bubbling of CO 2 20, addition of solvents 21, 22, or via external triggers including light 11 and temperature 23, 24, 25, 26. Thus, recent work has focussed on controlled release upon external stimuli 12. However, many applications, such as in biomedicine 8 or catalysis 3, 9, 10, 11 also require the release of the emulsified liquid. With particles strongly adsorbed to the interface, Pickering emulsions exhibit long-term stability, which makes them ideal for storage. Although discovered more than a century ago, they received renewed interest due to the desire to decrease the use of potentially environmentally damaging surfactants 3, 4, 5 and the increased abundance of particles able to adsorb at an oil–water interface 6, 7. Pickering emulsions are metastable dispersions of two immiscible liquids, kinetically stabilized by colloidal particles that partially wet both fluids 1, 2. This underscores the pivotal role of microgel morphology and the forces they exert on liquid interfaces in the control and design of stimuli-responsive emulsions and interfaces. Furthermore, microgels shared between two emulsion droplets in flocculated emulsions facilitate stimuli-responsiveness, regardless of their internal architecture. Conversely, the polymeric properties of the microgels enable them to spread and flatten at the liquid interface, enabling stimuli-responsive behaviour. However, limited deformability can lead to non-responsive emulsions. The colloidal properties of the microgels provide the foundation for the long-term stability of Pickering emulsions. Consequently, the stability of these emulsions is intricately tied to the nature of the stabilizing microgel particles - whether they are more polymeric or colloidal, and the morphology they assume at the liquid interface. 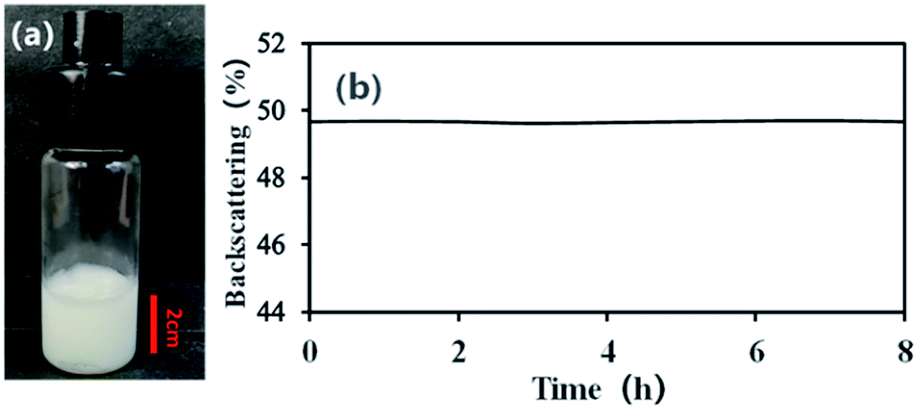
This study establishes that thermo-responsive emulsion behaviour is primarily determined by interactions between, rather than within, interfaces. Stimuli-responsive emulsions offer a dual advantage, combining long-term storage with controlled release triggered by external cues such as pH or temperature changes.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
